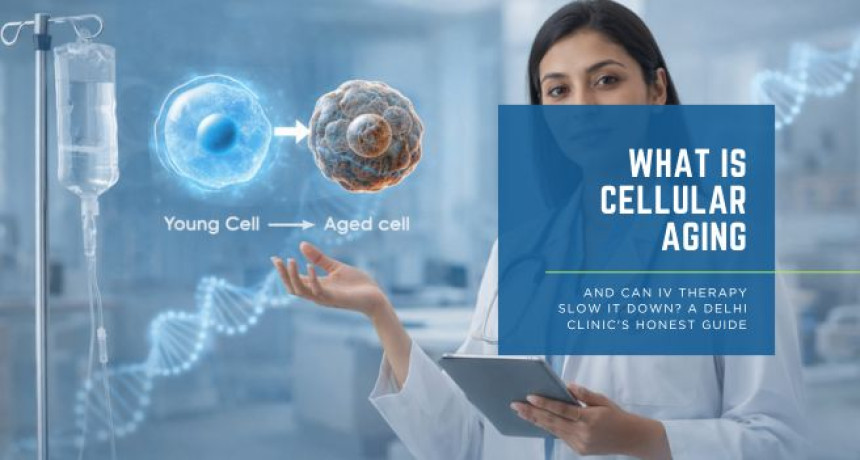
What Is Cellular Aging and Can IV Therapy Slow It Down? A Delhi Clinic's Honest Guide
2026-03-25 The anti-ageing industry is worth billions of dollars globally — and it is built, in significant part, on vagueness. Creams that "restore cellular youth." Supplements that "reverse the ageing clock." IV drips marketed with language designed to impress rather than inform. In this environment, the patient who wants an honest answer — what is actually happening inside ageing cells, and what can IV therapy genuinely do about it — is poorly served. This is that honest guide. At L&B Clinics, we believe that patients make better decisions when they understand the biology — not a simplified version of it, but the actual mechanisms that drive cellular ageing and the evidence that either supports or fails to support specific interventions. If you are considering IV therapy for longevity or anti-ageing purposes, you deserve to understand precisely what you are doing and why it may work. Here is the science. Here is the evidence. Here is the honest answer. Ageing is not a single event that happens to the body. It is the cumulative consequence of multiple, simultaneous biological processes occurring at the cellular level — processes that have been systematically identified, categorised, and studied over the past two decades of geroscience research. The most significant framework in modern longevity science is the hallmarks of ageing, first described by López-Otín et al. (2013) in Cell and updated in 2023 to include twelve distinct biological processes: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis. Each hallmark represents a specific molecular or cellular failure mode. Each contributes independently to the deterioration of function that we experience as ageing. And critically — each is measurable and modifiable to varying degrees through clinical intervention. Understanding which of these hallmarks IV therapy actually addresses, and through which mechanisms, is the foundation of an honest answer to whether it can slow cellular ageing. The Six Cellular Processes IV Therapy Directly Targets Mitochondria are the organelles responsible for producing ATP — the energy currency of every cell in the body. As cells age, mitochondrial function deteriorates progressively: mitochondrial membrane potential declines, reactive oxygen species production increases, and the efficiency of the electron transport chain falls. The result is reduced cellular energy output, increased oxidative damage, and the metabolic fatigue that is one of the most universal and recognisable features of biological ageing (Martínez-Reyes and Chandel, 2020). The primary driver of age-related mitochondrial dysfunction is the decline in NAD+ — the coenzyme that sits at the centre of mitochondrial energy metabolism and serves as the substrate for the sirtuin proteins that regulate mitochondrial biogenesis, antioxidant defence, and stress resistance. NAD+ levels fall by approximately 50 percent between the ages of 40 and 60, and this decline is now established across multiple peer-reviewed studies as a causal contributor to mitochondrial dysfunction rather than simply a correlate of it (Verdin, 2015). IV NAD+ therapy directly restores the substrate that mitochondrial function depends upon. It does not require enzymatic conversion, does not face gastrointestinal absorption variability, and achieves plasma concentrations in a single session that oral supplementation cannot reliably produce over weeks of use. Every cell in the body generates free radicals as a by-product of normal metabolism. Under healthy conditions, the body's antioxidant systems — primarily glutathione, vitamin C, vitamin E, and the superoxide dismutase enzyme family — neutralise these free radicals before they can cause significant damage. With age, free radical production increases while antioxidant capacity declines, producing the oxidative stress that damages DNA, proteins, and lipid membranes throughout the body (Pizzorno, 2014). In Delhi's urban environment, this process is significantly accelerated. PM2.5 air pollution generates systemic oxidative stress through multiple pathways — direct cellular toxicity, pulmonary inflammation, and systemic immune activation — that deplete glutathione and vitamin C at rates considerably higher than in less polluted environments. IV glutathione and high-dose vitamin C address this hallmark directly and with strong mechanistic support. Glutathione operates as the primary intracellular antioxidant, directly neutralising reactive oxygen species and regenerating other antioxidants after their oxidative cycles. Vitamin C operates in the extracellular space and serves as the electron donor that recycles oxidised glutathione back to its active reduced form. Together, they restore the antioxidant network comprehensively — at concentrations that oral supplementation cannot achieve and that are directly relevant to the oxidative load of urban Indian life (Weschawalit et al., 2017). The epigenome is the system of molecular switches that regulates which genes are expressed in each cell and when. As cells age, the epigenome undergoes progressive dysregulation — methylation patterns shift, histone modifications alter gene accessibility inappropriately, and the precision of gene expression that characterises young, healthy tissue is progressively lost. This epigenetic drift is now measurable through DNA methylation clocks — tools that generate a biological age estimate from the pattern of methylation across thousands of genomic sites (Horvath, 2013). Vitamin C's role in epigenetic maintenance is among the most significant and underappreciated findings in recent longevity science. It is an essential cofactor for the TET family of dioxygenase enzymes — the molecular machinery that performs DNA demethylation and maintains appropriate epigenetic programming. Cimmino et al. (2018) demonstrated that TET enzyme activity is directly dependent on vitamin C availability, establishing that adequate vitamin C at the concentrations achievable only through IV delivery is a prerequisite for resisting age-related epigenetic drift. NAD+ contributes to epigenetic maintenance through sirtuin activation. SIRT1 and SIRT6 — NAD+-dependent deacetylases — regulate histone modifications, DNA repair, and transcriptional programmes central to healthy ageing. Their activity falls directly with NAD+ decline, linking mitochondrial energy metabolism and epigenetic regulation through a single molecular hub (Verdin, 2015). Cellular senescence is the process by which cells that have sustained DNA damage or reached their replicative limit enter a permanent state of cell cycle arrest. Senescent cells do not die — they remain metabolically active and secrete a cocktail of pro-inflammatory cytokines, proteases, and growth factors collectively termed the senescence-associated secretory phenotype (SASP). The chronic low-grade inflammation driven by the accumulation of senescent cells — now termed inflammaging — is a primary driver of virtually every age-related disease, including cardiovascular disease, neurodegeneration, metabolic syndrome, and cancer (Franceschi et al., 2018). Glutathione and alpha-lipoic acid both suppress NF-κB signalling — the master transcriptional regulator of inflammatory cytokine production — reducing the inflammatory output of senescent cells and the systemic inflammaging burden that accelerates cellular ageing throughout the body. Magnesium, consistently depleted in Indian summer conditions and in stress, is a direct physiological antagonist of inflammatory signalling through calcium channel regulation (Rosanoff, Weaver and Rude, 2012). DNA damage accumulates continuously throughout life from UV radiation, oxidative stress, environmental toxins, and normal metabolic by-products. The efficiency with which cells detect and repair this damage determines, in large part, how rapidly genomic instability accumulates and how quickly age-related dysfunction develops. DNA repair is an enzymatic process that requires specific nutrients as cofactors at every step. NAD+ is the substrate for PARP-1, the primary enzyme responsible for detecting single-strand DNA breaks and initiating the repair response. B vitamins — particularly B12, folate, and B6 — are essential for the one-carbon metabolism that produces the nucleotide precursors required to fill repaired DNA sequences. Zinc is a structural component of multiple DNA repair enzymes, including the zinc finger proteins that read and bind specific DNA sequences during the repair process (Ames, 2018). IV therapy formulations that combine NAD+, B-complex, and zinc simultaneously restore the complete biochemical toolkit of DNA repair — addressing genomic instability at the substrate level rather than through any single molecular pathway. The nutrient sensing pathways — primarily the mTOR, AMPK, insulin/IGF-1, and sirtuin signalling networks — function as the cell's metabolic intelligence system, calibrating energy production, protein synthesis, cellular maintenance, and stress responses to the availability of nutrients and energy. With age, these pathways become progressively dysregulated: mTOR signalling becomes constitutively overactive, suppressing the autophagy processes that clear damaged cellular components; AMPK sensitivity declines, reducing mitochondrial biogenesis; sirtuin activity falls with NAD+ decline (López-Otín et al., 2013). Alpha-lipoic acid activates AMPK directly — mimicking the cellular signal of energy depletion that triggers mitochondrial biogenesis, autophagy, and metabolic recalibration. NAD+ restoration re-engages sirtuin activity, rebalancing the entire nutrient sensing network. These interventions do not simply add nutrients — they recalibrate the signalling environment through which cells govern their own maintenance and longevity (Shay et al., 2009). An honest guide requires this section. IV therapy does not reverse ageing. No currently available clinical intervention does. The hallmarks of ageing are deeply embedded biological processes that have been accumulating across decades — they cannot be eliminated in a clinical session or even a structured protocol of sessions. What IV therapy can do — when formulated correctly, administered in the right clinical context, and integrated into a broader lifestyle and medical framework — is meaningfully slow the progression of specific hallmarks, restore the nutritional and molecular substrates that cellular maintenance mechanisms depend upon, and reduce the physiological burden of ageing sufficiently to produce measurable improvements in energy, cognitive function, physical capacity, skin health, and inflammatory status. It is also worth being explicit about what the evidence actually shows. Most clinical research on NAD+, NMN, and related interventions is relatively recent and conducted primarily in animal models or small human cohorts. The mechanistic evidence is strong. The long-term clinical outcome data in humans is still developing. At L&B Clinics, we do not present IV therapy as a proven life extension intervention — we present it as an evidence-informed clinical tool for optimising the biological environment in which your own cellular repair and maintenance systems operate. That is a meaningful and genuinely valuable thing. It is not the same as reversing ageing, and conflating the two does a disservice to patients. Longevity IV therapy at L&B Clinics is appropriate for adults who are experiencing the functional markers of accelerated biological ageing — persistent fatigue that rest does not resolve, cognitive fog, reduced physical capacity and recovery, visible skin deterioration, sleep disruption, increased inflammatory symptoms, or metabolic changes — and who want to address these through a clinically supervised, evidence-informed protocol rather than unsupervised supplementation or wellness trends. Every patient begins with a clinical assessment. Where advanced biomarker testing is indicated — NAD+ metabolite levels, inflammatory markers, oxidative stress markers, micronutrient panels, or epigenetic biological age testing — these are discussed and recommended before any protocol is designed. The therapy follows the data, not a fixed menu. The goal is not a single dramatic session. It is a progressive, measurable improvement in biological age markers over a structured protocol — documented, assessed, and refined as the data evolves. References Ames, B.N. (2018) 'Prolonging healthy aging: longevity vitamins and proteins', Proceedings of the National Academy of Sciences, 115(43), pp. 10836–10844. https://doi.org/10.1073/pnas.1809045115 Cimmino, L., Dolgalev, I., Wang, Y., Yoshimi, A. and Aifantis, I. (2018) 'Restoration of TET2 function blocks aberrant self-renewal and leukemia progression', Cell, 170(6), pp. 1079–1095. https://doi.org/10.1016/j.cell.2017.07.032 Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. and Santoro, A. (2018) 'Inflammaging: a new immune-metabolic viewpoint for age-related diseases', Nature Reviews Endocrinology, 14(10), pp. 576–590. https://doi.org/10.1038/s41574-018-0059-4 Horvath, S. (2013) 'DNA methylation age of human tissues and cell types', Genome Biology, 14(10), p. R115. https://doi.org/10.1186/gb-2013-14-10-r115 López-Otín, C., Blasco, M.A., Partridge, L., Serrano, M. and Kroemer, G. (2013) 'The hallmarks of aging', Cell, 153(6), pp. 1194–1217. https://doi.org/10.1016/j.cell.2013.05.039 Martínez-Reyes, I. and Chandel, N.S. (2020) 'Mitochondrial TCA cycle metabolites control physiology and disease', Nature Communications, 11(1), p. 102. https://doi.org/10.1038/s41467-019-13668-3 Padayatty, S.J., Sun, H., Wang, Y., Riordan, H.D. and Levine, M. (2004) 'Vitamin C pharmacokinetics: implications for oral and intravenous use', Annals of Internal Medicine, 140(7), pp. 533–537. https://doi.org/10.7326/0003-4819-140-7-200404060-00010 Pizzorno, J. (2014) 'Glutathione!', Integrative Medicine: A Clinician's Journal, 13(1), pp. 8–12. Pullar, J.M., Carr, A.C. and Vissers, M.C.M. (2017) 'The roles of vitamin C in skin health', Nutrients, 9(8), p. 866. https://doi.org/10.3390/nu9080866 Rosanoff, A., Weaver, C.M. and Rude, R.K. (2012) 'Suboptimal magnesium status in the United States: are the health consequences underestimated?', Nutrition Reviews, 70(3), pp. 153–164. https://doi.org/10.1111/j.1753-4887.2011.00465.x Shay, K.P., Moreau, R.F., Smith, E.J., Smith, A.R. and Hagen, T.M. (2009) 'Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential', Biochimica et Biophysica Acta, 1790(10), pp. 1149–1160. https://doi.org/10.1016/j.bbagen.2009.07.026 Verdin, E. (2015) 'NAD+ in aging, metabolism, and neurodegeneration', Science, 350(6265), pp. 1208–1213. https://doi.org/10.1126/science.aac4854 Weschawalit, S., Thongthip, S., Phutrakool, P. and Asawanonda, P. (2017) 'Glutathione and its antiaging and antimelanogenic effects', Clinical, Cosmetic and Investigational Dermatology, 10, pp. 147–153. https://doi.org/10.2147/CCID.S128339 Yoshino, M., Yoshino, J., Kayser, B.D., Patti, G.J. and Imai, S. (2021) 'Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women', Science, 372(6547), pp. 1224–1229. https://doi.org/10.1126/science.abe9985What Cellular Ageing Actually Is
Mitochondrial Dysfunction
Oxidative Stress and Free Radical Accumulation
Epigenetic Alterations
Cellular Senescence and Systemic Inflammation
Genomic Instability and DNA Repair Impairment
Deregulated Nutrient Sensing
What IV Therapy Cannot Do — The Honest Part
Who Is a Candidate for Cellular Anti-Ageing IV Therapy at L&B Clinics
.png)